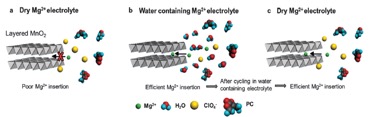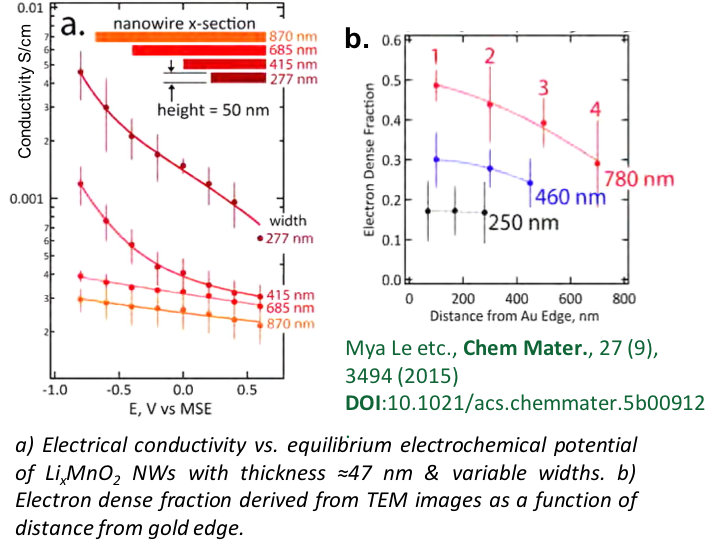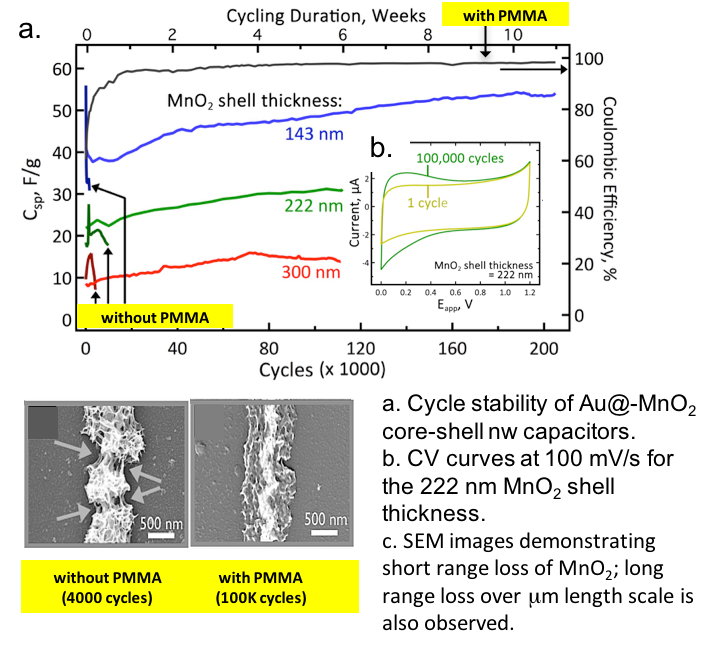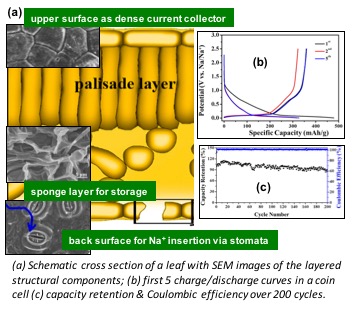
Predicting Dynamic Potential Distributions and Space-Charge Layers in Solid State Battery Systems
Scientific Achievement
- Established a theoretical framework to predict interface potential profiles from thermodynamic driving forces, defect formation energies, and electronic band alignments.
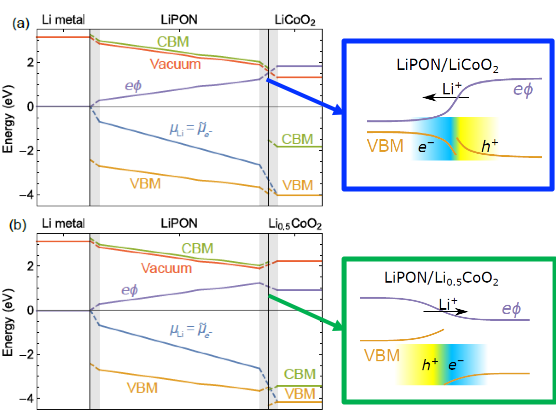
- Predicted that e- and Li+ transfer at LiPON/LixCoO2 interface depends on cathode state-of-charge, leading to field reversal from x=0.5 to x=1 and an explanation for conflicting experimental results.
Significance and Impact
Theoretical approach integrates behavior of electron-related voltages and ion-related chemical potentials in battery systems as transported ions alter electrode materials, changing interfacial space charge fields that modulate both ion and electron transport.
Research Details
- Chemical potential changes as lithiation proceeds have profound consequences for charge transport.
- Differing x=[Li] may explain different Li+ transport direction at LiPON/LixCoO2 inferred from experiment.
- Results suggest preferred solid electrolyte materials to increase discharge power.
References
- A.Michael W. Swift and Yue Qi, “First-principles prediction of potentials and space-charge layers in all-solid-state batteries”, Phys Rev Letters (in revision).
Summary
Solid state batteries, differentiated by their use of solid electrolytes, offer advantages in safety (no flammable liquid electrolyte) and in the 3D structures that can be achieved. When fabricated in thin film form, a wide variety of battery architectures and form factors are possible. As example, battery materials can be configured in 3D interpenetrating-electrode architectures that promise simultaneous energy-power performance, along with long cycle life and safety. But while thin film fabrication enables high power at high energy, it also places a premium on understanding and control of solid state electrochemical interfaces (e.g., electrode/solid-electrolyte)
This raises a profound scientific challenge to understand the coupled behavior of ions and electrons at solid state electrochemical interfaces. The battery community has long studied ion transport in liquid electrolyte systems and battery electrodes, with electrons playing a secondary role. At the same time the solid state electronics community has investigated many aspects of electron transport and interfaces, but in the absence of mobile ions. Solid state battery operation relies on coupled, interdependent ion and electron behavior, posing a conceptual and theoretical challenge to address in a self-consistent fashion. As a heterojunction system, solid state batteries provide a landscape of energy bands and barriers that affect not only electron transport but also the ion transport on which battery function is based. However, the intrinsic functionality of charge/discharge alters the electrode materials themselves, e.g. through lithiation/delithiation, and when the materials change their energy bands and barriers change as well - hence the need to address ion and electron behavior together in a coherent framework.
This highlight presents a fully first-principles-informed model of potential profiles in a model solid state battery system, Li-anode/LiPON-solid-electrolyte/LixCoO2-cathode. The profiles account for thermodynamic driving forces, defect formation energies, and electronic band alignments. The approach integrates behavior of electron-related voltages and ion-related chemical potentials and shows that - contrary to purely electronic systems - the electronic Fermi level changes to drive battery operation. An unexpected prediction is that the direction of the electric field driving Li transport at the LiPON/LixCoO2 interface reverses as the cathode state-of-charge x changes from 0.5 to 1.0. The changing direction of Li flow, dependent on x, may explain conflicting conclusions drawn from experiments. These observations suggest several ways that could be effective in reducing interface resistance in solid state batteries.
Acknowledgements
This work was supported by the NEES-EFRC.